More Info
Script
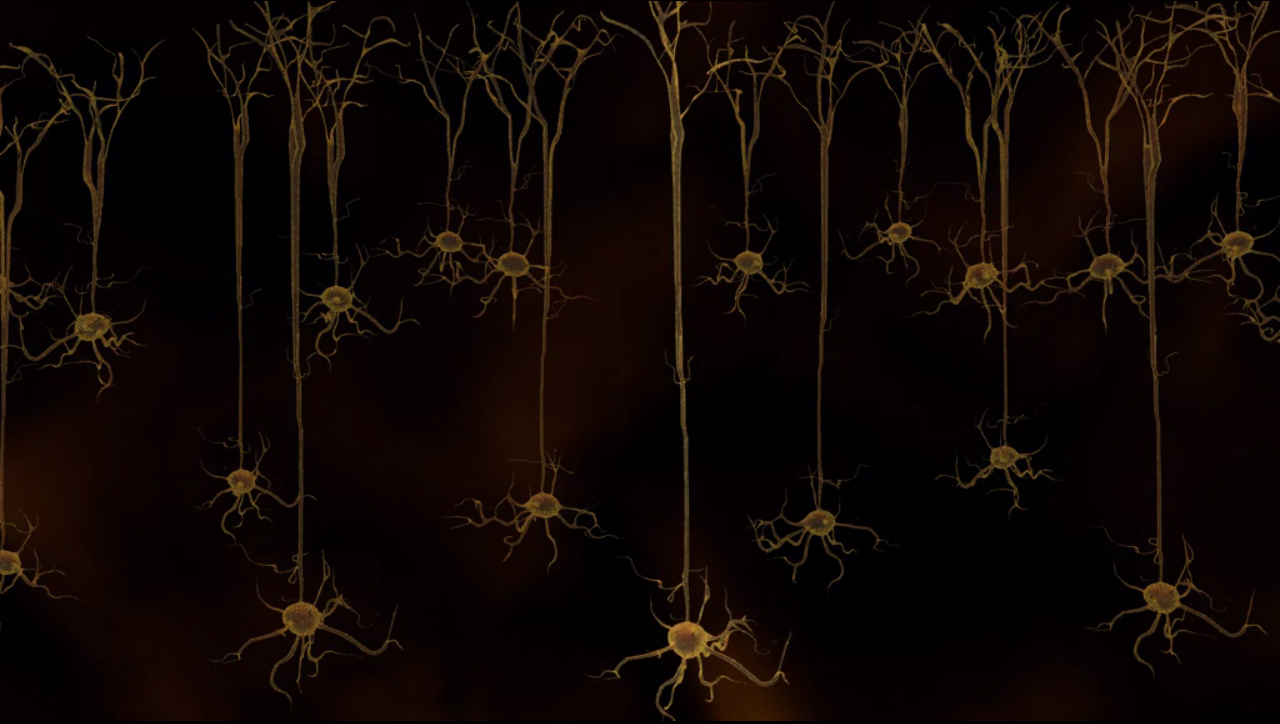
What if we could control brain activity with the ease and speed of a light switch? Scientists have done just that using light-activatable molecules produced by organisms such as green algae and bacteria. These molecules are cell-membrane channels called opsins, and they respond to light of specific wavelengths by allowing the selective passage of ions, which in brain cells would translate into changes in electrical activity: the currency of communication in the brain. The challenge is to introduce these opsins into animal cells. Scientists have accomplished this feat thanks to the vast arsenal of molecular tools.
First, we need to identify and isolate the genes that encode for the opsin.
Next, we use colonies of the bacteria E. Coli as factories to produce many copies of the opsin genes. Only colonies expressing the opsin genes survive and are subsequently harvested, digested and purified for the opsin genes.
After we have large quantities of the opsin genes, we incorporate them into viruses that specialize in infecting neurons.
The viral suspension is then injected directly into the brain of an animal, a mouse being a typical subject. The viruses can be engineered to infect specific types of neurons, adding powerful selectivity to the optogenetic manipulation of brain activity.
In the end, small groups of neurons in the mouse brain are genetically modified to be sensitive to light. Now we can finally exploit these light-activatable channels to gain a better understanding of brain function and dysfunction, and explore therapeutic applications.
To deliver light to opsin-expressing neurons, we use LEDs or thin optical fibers. Ultimately, the impact of light-activation on neural activity will depend on the opsin of choice.
For example:
To reduce neural activity, we insert Halorhodopsin into neurons. Yellow light opens Halorhodopsin channels on a neuron’s membrane and allows the entry of chloride ions, which inhibit the generation of action potentials by neurons.
To increase neural activity, we insert Channelrhodopsin into neurons. Blue light opens Channelrhodopsin channels on a neuron’s membrane and allows the entry of cations, which depolarize neurons inducing more action potentials.
Optogenetics also has the potential to alleviate the symptoms of neurodegenerative diseases such as Parkinson. Elderly patients experience chronic tremors and if we can pinpoint the population of neurons responsible for this neurological disorder and whose electrical activity has gone awry, we can insert light-activatable channels to correct their activity. Optogenetics may not cure diseases, but it can potentially improve quality of life.
First, we need to identify and isolate the genes that encode for the opsin.
Next, we use colonies of the bacteria E. Coli as factories to produce many copies of the opsin genes. Only colonies expressing the opsin genes survive and are subsequently harvested, digested and purified for the opsin genes.
After we have large quantities of the opsin genes, we incorporate them into viruses that specialize in infecting neurons.
The viral suspension is then injected directly into the brain of an animal, a mouse being a typical subject. The viruses can be engineered to infect specific types of neurons, adding powerful selectivity to the optogenetic manipulation of brain activity.
In the end, small groups of neurons in the mouse brain are genetically modified to be sensitive to light. Now we can finally exploit these light-activatable channels to gain a better understanding of brain function and dysfunction, and explore therapeutic applications.
To deliver light to opsin-expressing neurons, we use LEDs or thin optical fibers. Ultimately, the impact of light-activation on neural activity will depend on the opsin of choice.
For example:
To reduce neural activity, we insert Halorhodopsin into neurons. Yellow light opens Halorhodopsin channels on a neuron’s membrane and allows the entry of chloride ions, which inhibit the generation of action potentials by neurons.
To increase neural activity, we insert Channelrhodopsin into neurons. Blue light opens Channelrhodopsin channels on a neuron’s membrane and allows the entry of cations, which depolarize neurons inducing more action potentials.
Optogenetics also has the potential to alleviate the symptoms of neurodegenerative diseases such as Parkinson. Elderly patients experience chronic tremors and if we can pinpoint the population of neurons responsible for this neurological disorder and whose electrical activity has gone awry, we can insert light-activatable channels to correct their activity. Optogenetics may not cure diseases, but it can potentially improve quality of life.
Subject
Optogenetics, opsin, parkinson's disease, neurons, excitatory, inhibitory, light-activated cell-membrane channels, Channelrhodopsin, Halorhodopsin
Type
Animation
Format
MPEG-4
Date
03/21/2016
Contributor
Ching-Jung Chen, Katie Cheng, Timmy Eng, Rafay Malik, Hysell Oviedo, Anna Cahn, Advait Apte
Publisher
The City College Libraries, New York, New York
Identifier
ANI009
Language
English
Rights
Optogenetics by City College of New York Digital Scholarship Services is licensed under a Creative Commons Attribution 4.0 International License.